|
Where #M# is the molar mass in grams per mole ( #gmol^-1#), #m# is the mass of the sample in grams ( #g#), and #n# is the amount in moles of the sample ( #mol#). Every hydrogen atom has one proton in its nucleus. Hydrogen, at the upper left of the table, has an atomic number of 1. In this table, an elements atomic number is indicated above the elemental symbol. Ask your teacher or tutor if you are not sure which one to use.Īnother way to find molar mass if amount and mass of a substance are known is by using the formula The periodic table (see figure below) displays all of the known elements and is arranged in order of increasing atomic number. 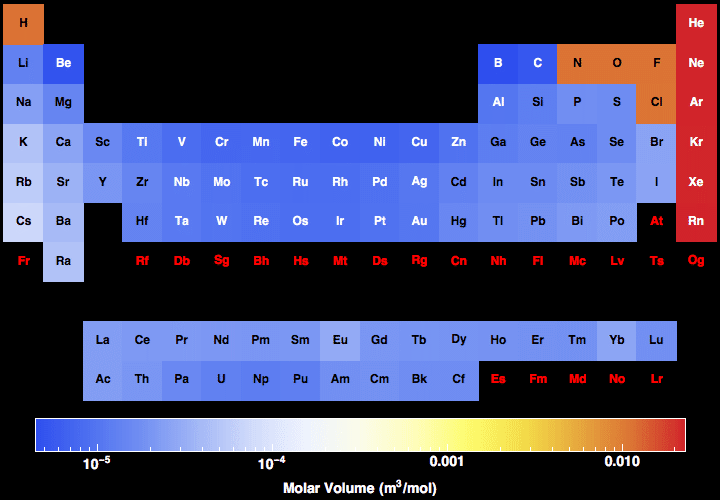
The molar masses will be slightly different if you are using a table with more accurate decimal values for atomic mass. Sulfuric acid #H_"2"SO_4# has 2 Hydrogen atoms (mass #1#), 1 sulfur atom (mass #32#) and 4 oxygen atoms (mass #16#) so: For elements with no stable isotopes, the mass number of the isotope with the longest half-life is in parentheses. Then simply find out how many of each atom is in the molecule and add the respective atomic masses to find the molecular or molar mass.įor example water #H_2O# has 2 Hydrogen atoms (mass #1#) and 1 oxygen atom (mass #16#) so: I prefer to use tables with whole number atomic masses, but that is just personal preference. Please note that the elements do not show their natural relation towards each other as in the Periodic system. The unity for atomic mass is gram per mol. 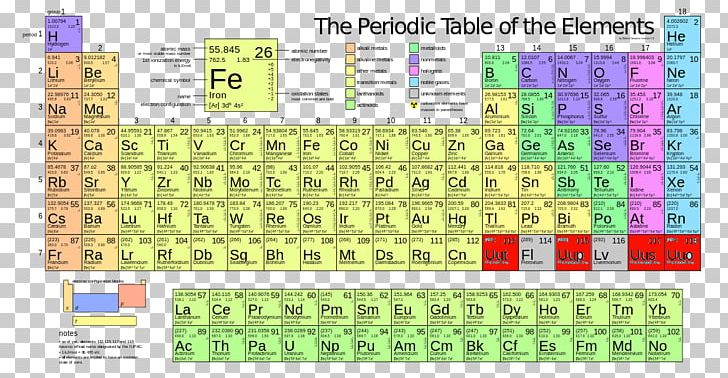
The lightest chemical element is Hydrogen and the heaviest is Hassium. Find a periodic table and look at the atomic mass of each atom in the molecule. For chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
